In this case the principal quantum numbers are 6, 5, and 4. In lanthanum, as a further illustration, the electrons involved are in the 6s 5d and 4f orbitals, respectively. the electron that differentiates an element from the previous one), or the differentiating electron according to the aufbau approximation. For example, the highest occupied orbital electron, the actual differentiating electron (i.e. Thus, a complete description of the system can be given with fewer quantum numbers, if orthogonal choices are made for these basis vectors.ĭifferent electrons in a system will have different quantum numbers. The spin–orbital interaction, however, relates these numbers. 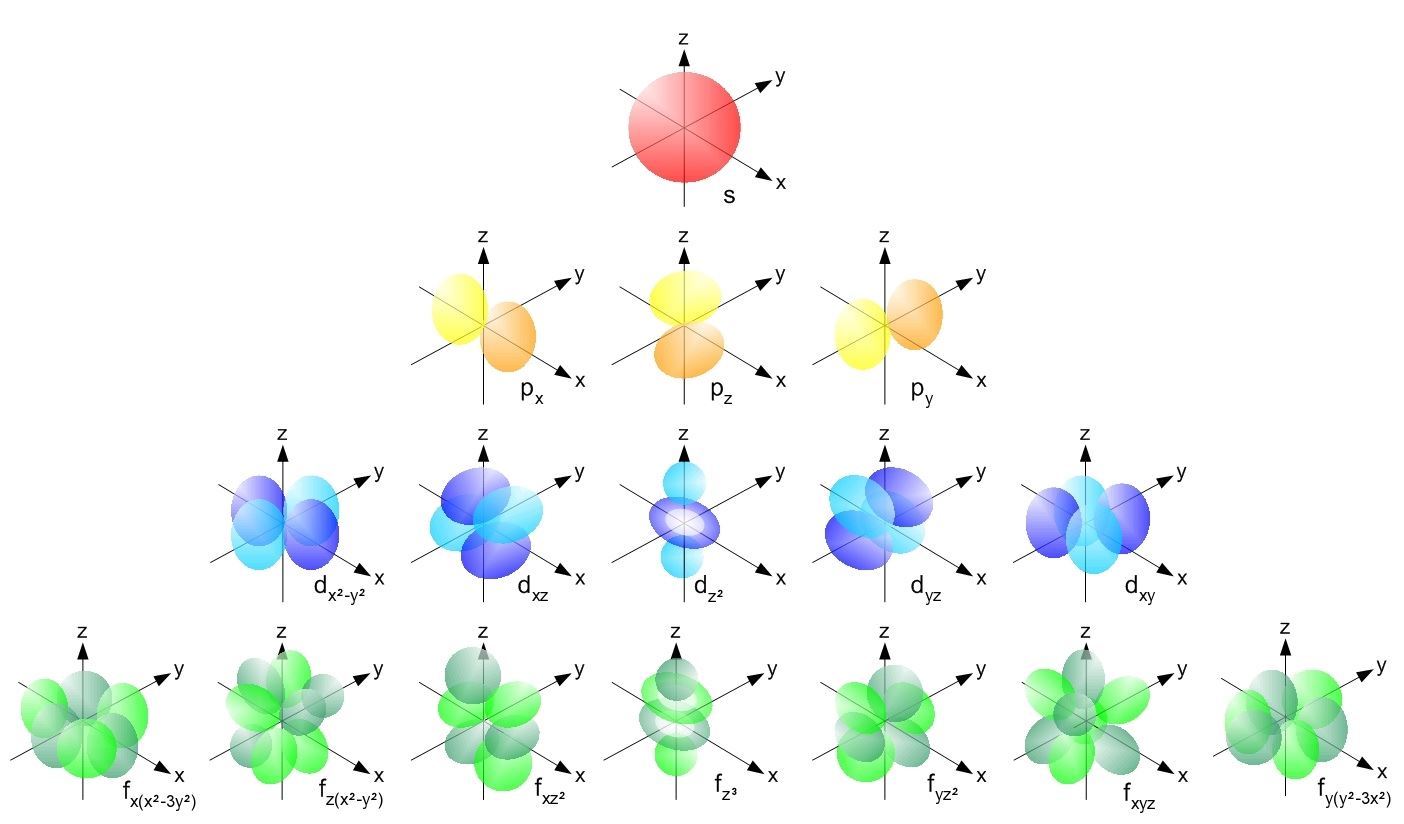
As a result of the different basis that may be arbitrarily chosen to form a complete set of commuting operators, different sets of quantum numbers may be used for the description of the same system in different situations.įour quantum numbers can describe an electron in an atom completely: There is a one-to-one relationship between the quantum numbers and the operators of the CSCO, with each quantum number taking one of the eigenvalues of its corresponding operator. A complete set of commuting observables (CSCO) that commute with the Hamiltonian characterizes the system with all its quantum numbers. There is also one quantum number for each linearly independent operator O that commutes with the Hamiltonian. 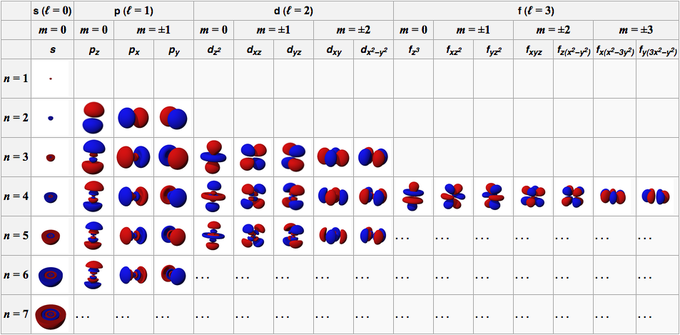
There is one quantum number of the system corresponding to the system's energy i.e., one of the eigenvalues of the Hamiltonian. time evolution) of any quantum system are described by a quantum operator in the form of a Hamiltonian, H. A quantized system requires at least one quantum number. Hence these parameters must be found for each system to be analyzed. The tally of quantum numbers varies from system to system and has no universal answer. Quantum numbers needed for a given system Any quantum system can have one or more quantum numbers it is thus difficult to list all possible quantum numbers. 
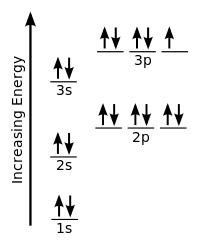
An important family is flavour quantum numbers – internal quantum numbers which determine the type of a particle and its interactions with other particles through the fundamental forces. Quantum numbers often describe specifically the energy levels of electrons in atoms, but other possibilities include angular momentum, spin, etc. This distinguishes quantum mechanics from classical mechanics where the values that characterize the system such as mass, charge, or momentum, all range continuously. In particular, this leads to quantum numbers that take values in discrete sets of integers or half-integers although they could approach infinity in some cases. Together, a specification of all of the quantum numbers of a quantum system fully characterize a basis state of the system, and can in principle be measured together.Īn important aspect of quantum mechanics is the quantization of many observable quantities of interest. Quantum numbers correspond to eigenvalues of operators that commute with the Hamiltonian-quantities that can be known with precision at the same time as the system's energy -and their corresponding eigenspaces. List all the allowed combinations of the four quantum numbers ( n, l, m l, m s) for electrons in a 2 p orbital and predict the maximum number of electrons the 2 p subshell can accommodate.In quantum physics and chemistry, quantum numbers describe values of conserved quantities in the dynamics of a quantum system. An electron behaves like a magnet that has one of two possible orientations, aligned either with the magnetic field or against it. These are described by a fourth quantum number ( m s), which for any electron can have only two possible values, designated +½ (up) and −½ (down) to indicate that the two orientations are opposites the subscript s is for spin. In an external magnetic field, the electron has two possible orientations (Figure 6.7.2). In a magnetic field, an electron has two possible orientations with different energies, one with spin up, aligned with the magnetic field, and one with spin down, aligned against it. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
